The most relevant medtech news and market signals this week for commercial, digital, and strategy leaders across Europe.
This week’s Europe medtech news points to a market that is becoming less patient with vague AI platform stories and more interested in deployment readiness: products that fit workflows, evidence plans that survive scrutiny, and policy moves that reduce friction instead of adding more theatre.
For founders, operators, and investors, that is the real signal. Capital is still available, but the market is rewarding commercialization discipline, not novelty alone.
Executive memo
If I were reading this as a CEO, GM, or VP Commercial, I would take away one thing: the market is rewarding products that are easier to imagine inside a budget, a workflow, and a compliance plan.
Waiv and Vitestro fit that pattern from different angles. One sits close to precision-testing infrastructure and trial utility. The other tackles a painful clinical workflow with clearer deployment logic. EIT Health’s validation funding and the latest EU AI simplification push reinforce the same point: evidence and compliance still cost money, but Europe is starting to acknowledge that scale breaks when validation and regulation become pure drag.
Money Flows
Waiv raises $33M to scale AI-powered precision testing
What happened
Owkin spun out Waiv, formerly Owkin Dx, with $33M in financing led by OTB Ventures and Alpha Intelligence Capital.
Why it matters
This is not a generic diagnostics AI. It is a bet on precision-testing infrastructure that can influence patient stratification in care and clinical trials, which gives it a clearer route to budget ownership and enterprise relevance.
What it may signal
From a commercialization perspective, capital still flows to AI when it is tied to evidence generation, trial utility, and workflow fit rather than broad platform storytelling.

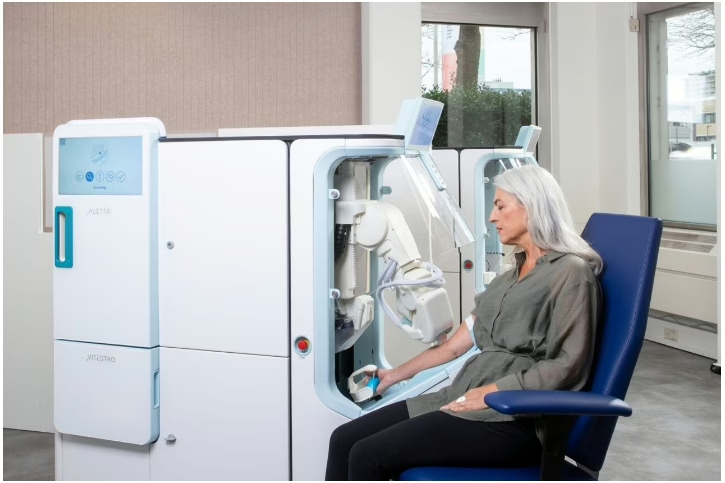
Vitestro secures $70M Series B for autonomous blood collection
What happened
Vitestro raised an oversubscribed $70M Series B to advance development and commercialization of Aletta, its autonomous blood-drawing robot. The company already holds CE mark and is using Europe as a proving ground while preparing for the US.
Why it matters
Hospitals do not buy robotics because robotics are exciting. They buy them when labor pressure is real, the task is repetitive, and deployment starts to look operationally credible.
What it may signal
For medtech commercial teams, automation stories will win faster when they are tied to specific workflow pain points and realistic implementation, not category hype.

Spotlight Pathology lands seed funding for AI in blood cancer diagnosis
What happened
Liverpool-based Spotlight Pathology raised £1.4M in seed funding to develop digital pathology software for blood cancer detection.
Why it matters
The round is modest, but the story is strong: a narrow use case, a clinically legible problem, and an NHS-relevant deployment path.
What it may signal
Investors still back early AI diagnostics when the adoption story is crisp, and the evidence burden looks manageable.

Signals to Watch
MiniMed expands its European diabetes ecosystem with a new CE-marked sensor pairing
MiniMed said it received CE mark approval for its MiniMed 780G system with the Instinct sensor from Abbott, with first commercial launches in European countries expected this summer.
Commercial lens: In diabetes, stickiness still comes from ecosystem depth and interoperability, not a single hardware update.

EIT Health opens validation funding for digital and AI medical technologies
EIT Health opened its 2026 Innovation Validation Call, offering up to €850,000 per project and up to 50% co-funding for late-stage validation work, including pivotal studies, real-world evidence, CE-mark submission support, product finalization, and market readiness.
Commercial lens: This is one of the most practical capital layers in Europe. It helps close the gap between prototype enthusiasm and procurement-ready reality.

Why this matters?
Commercially speaking
For founders, that means tighter positioning around workflow fit, evidence burden, and adoption logic. For commercial leaders, it means spending less time admiring innovation theatre and more time asking whether a product can survive procurement, integration, and reimbursement reality.
What leaders should watch next?
Watch whether European policy simplification creates real operating relief or just new layers of interpretation.
Watch which companies move from pilot language to procurement language. That is usually where the real market starts.
Strategic question for the C-suite
Europe MedTech News FAQ
The week’s biggest Europe medtech news included Waiv’s $33M financing, Vitestro’s $70M Series B, a new MiniMed CE mark, EIT Health validation funding, and fresh EU AI policy signals.
Waiv, Vitestro, and Spotlight Pathology were among the most notable funding stories in European medtech and digital health this week.
This week brought fresh pressure to simplify AI compliance in Europe and continued emphasis on validation and evidence readiness for digital and AI-powered medical technologies.
Get Europe MedTech News in your inbox
A weekly executive read on the most relevant medtech and digital health signals in Europe, with a focus on commercialization, evidence, and operating reality.
