Europe MedTech & Digital Health Weekly Brief #30
Week of March 23–29, 2026
This week was not really about invention. It was about who controls the operating layer: clinical evidence infrastructure, procedure-room workflow, diagnostics pathways, cyber recovery, and the regulatory plumbing that increasingly shapes commercial readiness.
Key Signals
- Clinical trial data infrastructure consolidated under a scaled platform owner.
- Philips pushed AI deeper into the procedure room and workflow closer to the point of care.
- Abbott strengthened its position in cancer screening and precision diagnostics.
- Stryker showed how fast cyber risk becomes a manufacturing and customer-service problem.
- EUDAMED moved one step closer from regulatory admin to commercial reality.
Executive Memo
Zoom out, and the pattern is operating-model resilience.
Thermo Fisher completed its acquisition of Clario, strengthening its position in clinical trial endpoint and evidence infrastructure. Philips received FDA clearance for DeviceGuide, bringing AI-assisted real-time guidance into minimally invasive heart valve repair, and also launched IntraSight Plus as an integrated coronary intervention platform. Abbott completed Exact Sciences, deepening its cancer diagnostics position. Stryker, meanwhile, showed that in MedTech, cyber disruption stops being a technical story the moment manufacturing and ordering are hit.
The common thread is simple: advantage in MedTech is increasingly built not only on product features, but on control of a workflow, a data layer, a care pathway, or a recovery capability that customers depend on. That is where commercial leverage is accumulating.
Signals to Watch
Thermo Fisher × Clario — clinical data infrastructure consolidates
Thermo Fisher completed its acquisition of Clario on March 24. The company said the deal strengthens its clinical trial technology and services offering, and reiterated expectations for first-year EPS contribution and longer-term synergies.
Why it matters
This is not a classic device story. It is an infrastructure story. Endpoint data capture, patient-reported outcomes, and trial workflow are becoming more strategically valuable as evidence generation grows more central to both drug and device economics.

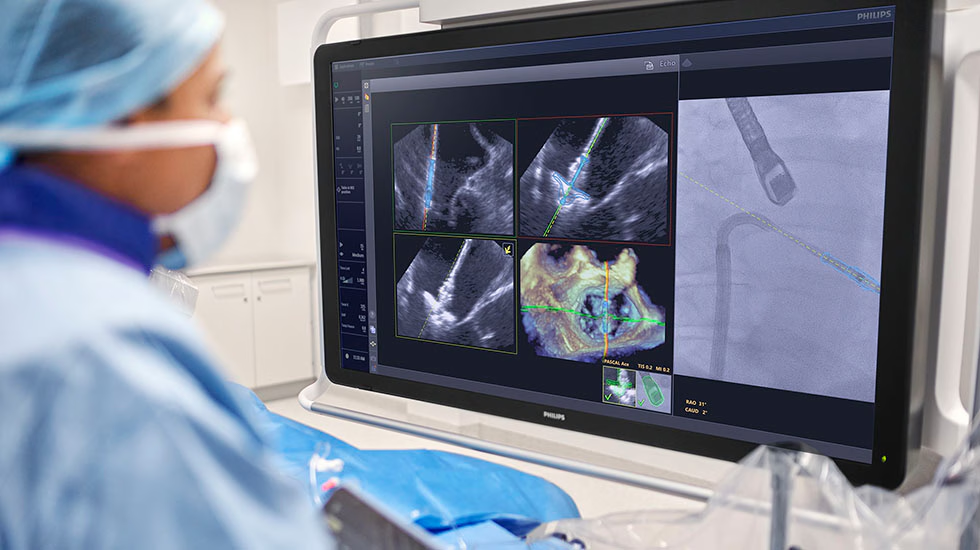
Philips — procedural AI moves closer to routine workflow
Philips received FDA 510(k) clearance for EchoNavigator R5.0 with DeviceGuide, intended for use with Edwards’ PASCAL Ace Mitral Valve Repair System, and said commercial availability will depend on market release and regulatory requirements. Philips also launched IntraSight Plus, a CE-marked and FDA-cleared coronary intervention platform.
Why it matters
This is a meaningful shift. It is not just AI interpreting an image after the fact. It is AI supporting navigation during the procedure itself, while integrated workflow platforms like IntraSight Plus tighten the connection between imaging, physiology, and treatment planning.
Abbott × Exact Sciences — diagnostics pathways get more concentrated
Abbott completed Exact Sciences on March 23, saying the acquisition strengthens its position in cancer screening and diagnostics.
Why it matters
This is not just scale for scale’s sake. It is a pathway move. The companies with broader positions across screening, monitoring, and treatment-decision support are becoming harder to dislodge.

Stryker — cyber resilience becomes a commercial capability
Stryker said most manufacturing sites and critical production lines were operating again after the March 11 cyberattack, and that its electronic ordering system had been restored.
Why it matters
For MedTech, cyber resilience is now visibly tied to manufacturing continuity, customer trust, and revenue protection. Once plants and orders are disrupted, the issue is no longer just technical. It is commercial.

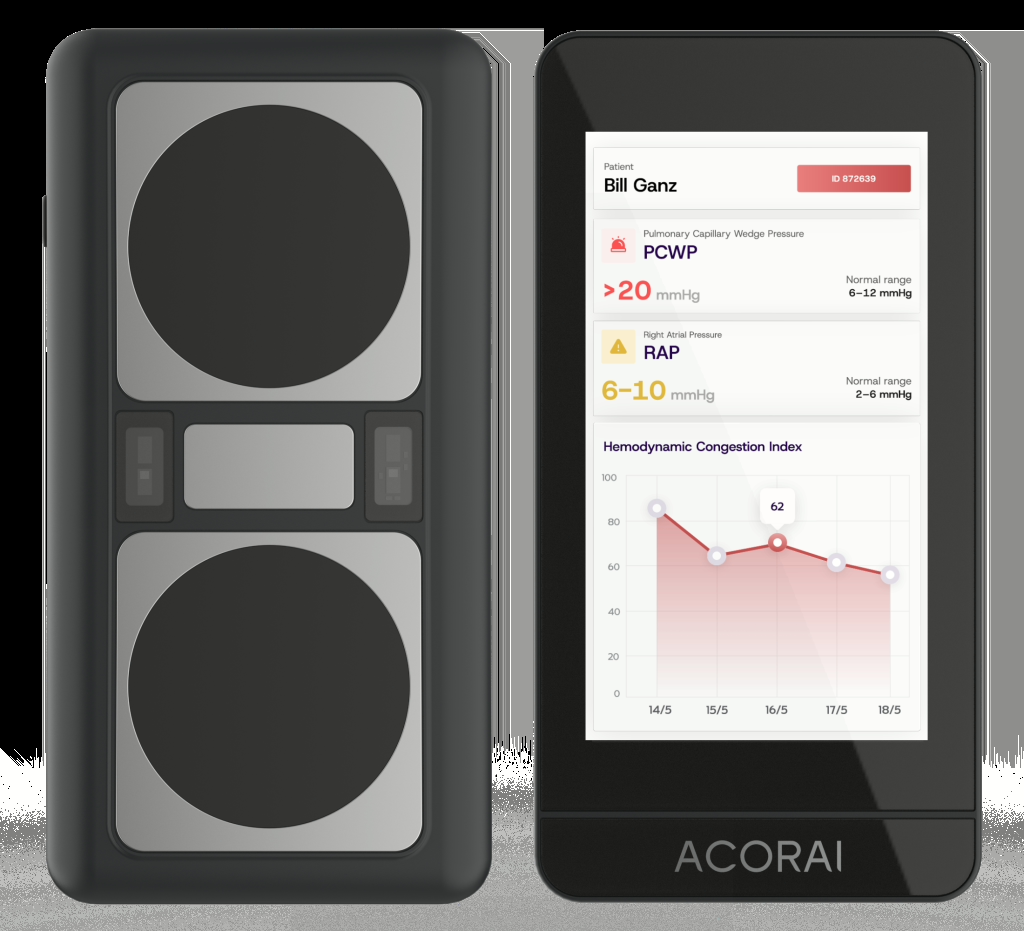
Acorai — validation milestone beats another funding headline
Acorai announced the initiation of its 1,000-patient HOSP-HF pivotal study and enrollment of the first patient, with study activity planned across Sweden, Denmark, Singapore, and the US during 2026.
Why it matters
That is the kind of smaller-company signal worth watching in 2026: less hype, more evidence. In this market, clinical validation milestones matter more than broad AI rhetoric.
People & Strategic Moves
Philips — John DeFord proposed for the Supervisory Board
Philips said it will propose John DeFord as a new Supervisory Board member at its 2026 AGM, alongside reappointments for Paul Stoffels, Herna Verhagen, and Sanjay Poonen.
Why it matters
This is not the biggest story of the week, but it is a clean governance signal. Philips is reinforcing its board with deeper MedTech, regulatory, and technology experience while pushing workflow integration and AI further into practice.

Mölnlycke — CEO transition
Mölnlycke announced on March 24 that CEO Zlatko Rihter will resign, with CFO Guillaume Joucla appointed interim CEO while the board begins the search for a permanent successor.
Why it matters
Leadership changes at this level are rarely random. In the current market, they read less like symbolic refreshes and more like signs of execution pressure and strategic discipline.

Regulatory and Policy Watch
EUDAMED — now close enough to affect readiness
Why it matters
This is crossing from regulatory administration into commercial readiness. Registration discipline and data completeness increasingly affect launch preparedness, distributor interactions, and procurement credibility.
EMA breakthrough-device pilot — one to watch in Q2
Why it matters
It is not the week’s biggest story, but it is worth tracking. Anything that improves the path for highly innovative devices in Europe matters for founders, investors, and regulatory leaders trying to judge whether the region can become less hostile to innovation at the margin.
AI Act × MDR/IVDR — use careful wording here
What Leaders Should Watch Next
Strategic Question for the C-Suite
Are you building something hospitals need to add to their workflow — or something that reduces friction inside a workflow they already cannot afford to slow down?
One Thing to Remember
In European MedTech right now, value is concentrating in the operating layer: clinical data infrastructure, procedure-room workflow, diagnostics pathways, regulatory readiness, and resilience under stress.
This content has been enhanced with AI.
Thermo Fisher, Philips, European Commission, EMA, Reuters, Abbott, Mölnlycke, Acorai.

